A New Era for Management of Medical Devices with Taiwan's Medical Devices Act to be Implemented on May 1st, 2021
The Legislative Yuan passed the third reading of the "Medical Devices Act” on December 13, 2019 and the Act was announced on January 15, 2020 under the Presidential Decree.
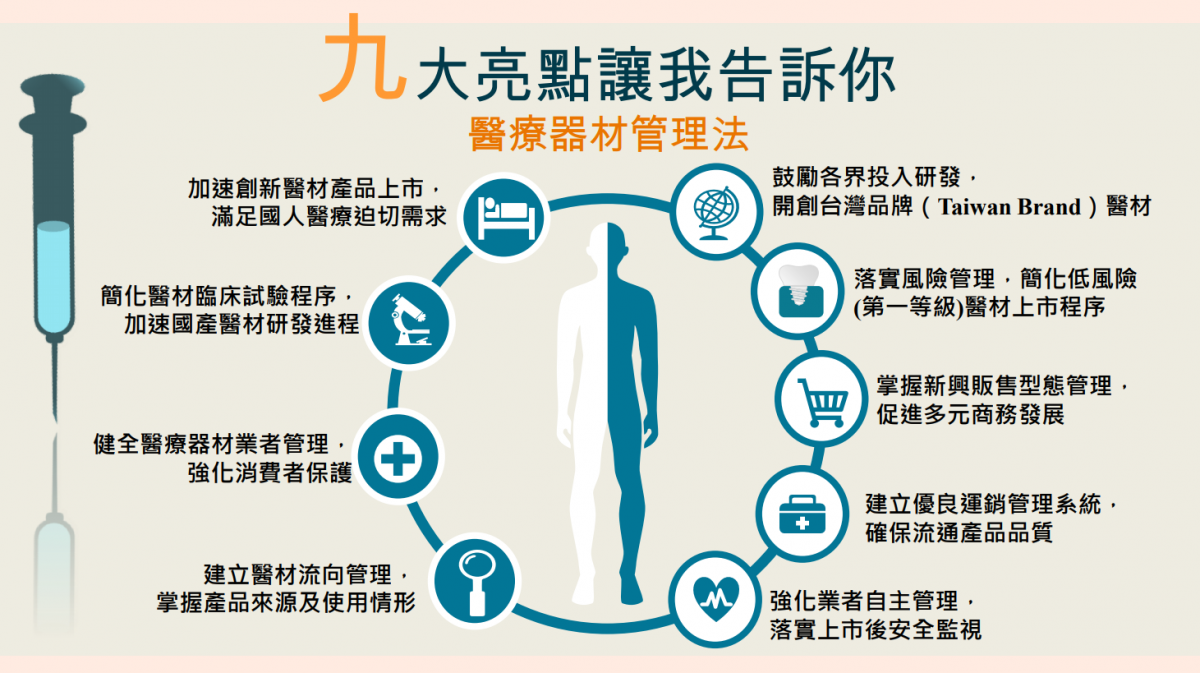
The Legislative Yuan passed the third reading of the “Medical Devices Act” on December 13, 2019 and the Act was announced on January 15, 2020 under the Presidential Decree. To ensure that medical devices firms will have sufficient time to make appropriate adjustments and also to ensure that the government has enough time to draft relevant regulations, the Executive Yuan has announced that the Medical Devices Act will take effect on May 1st, 2021. The Medical Devices Act will establish a system to effectively regulate medical devices throughout the medical device life cycle, marking a new start for medical device management in Taiwan.
Following world trends, Taiwan has amended the Pharmaceutical Affairs Act and stipulated a new law specifically for the management of medical devices. With the Medical Devices Act in place, Taiwan could establish an appropriate management system that takes the characteristics of medical devices into consideration. The new Act covers repair and maintenance of medical devices, sale and supply of medical devices, quality management system and distribution management of medical devices, electronic listing system for some low-risk products, flexible validity period for issuing licenses, medical device clinical trials, safety monitoring of medical devices, proactive reporting and more, to protect consumer safety and improve management of medical devices.
According to Taiwan Food and Drug Administration (TFDA), at present, TFDA has completed the drafting of relevant regulations and regulatory orders related to the Medical Devices Act, including Regulations Governing the Classification of Medical Devices, Regulations Governing Issuance of Medical Device License, Listing, and Annual Declaration, Regulations of Medical Device Tracking Management, and Regulations for Management of Medical Devices Technicians. The government has also introduced various measures to give medical device firms reasonable transition period to make adjustments and to minimize the impact to the industry.
Director-General Shou-Mei Wu of TFDA states that Taiwan’s medical device management system will continue to follow international trends. Taiwan will work to harmonize with international regulations and reduce regulatory barriers that Taiwanese medical device dealers face when they attempt to compete in the international market. At the same time, Taiwan will also work to protect consumers’ safety when they use medical devices, promote the development of the medical device industry, and enhance the competitiveness of medical device businesses in the international arena.
Date: 2021-05-01
Source: Food and Drug Administration, Ministry of Health and Welfare